Direct cyanation of aromatic rings using dinitrogen and methane
promoted by nonthermal plasma
Lei Yu+, Shixiong Zhang+, Liang Liu+, Di Li+, Yongli Cai, Chenxin Wu, Lei Hua, Haiyang Li, Dehui Deng, Ping Chen, Zhenfeng Xi*, Jianping Guo*, Junnian Wei*
Sci. Adv. 2025. DOI: 10.1126/sciadv.ady3414.
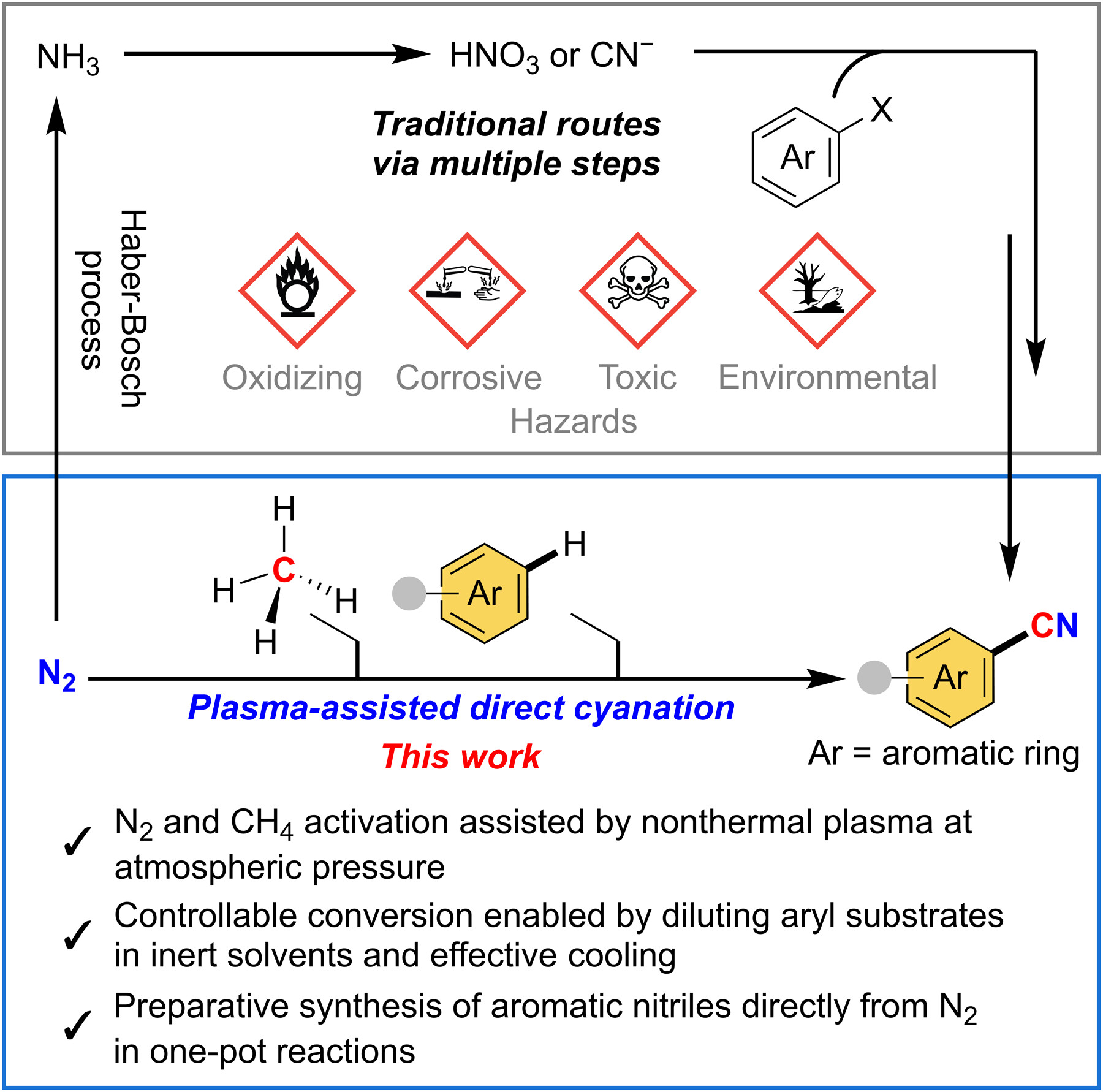
Directly converting dinitrogen (N2) into valuable nitrogen-containing compounds remains an enduring challenge in chemical synthesis. Here, we report the direct cyanation of aromatic substrates using N2 and methane (CH4) at atmospheric pressure facilitated by a custom-built, air-free dielectric barrier discharge (DBD) plasma system. A broad range of aromatic compounds, including benzene, were successfully transformed into their corresponding aromatic nitriles. Both experimental and computational evidence suggested that the reaction proceeds primarily via the in situ generation of •CN radicals from N2 and CH4 within the plasma zone. Subsequent radical addition to aromatic rings allowed the one-pot formation of aryl nitriles. This approach represents a major advancement in dinitrogen-based organic methodologies, providing an efficient alternative to conventional cyanation methods that heavily rely on lengthy synthetic routes and hazardous cyanide reagents.